Soil Chemistry
Before spending money on nutrients, make sure you have adjusted your pH to be optimal. Money spent on nutrients that the plant cannot uptake because of imbalanced soil chemistry is an investment with little to no return on investment. Start with pH, and let the soils work for you.
The pH scale ranges between 0 and 14, where pH 7 is neutral, pH values below 7 are acidic, and pH values above 7 are alkaline. The lower the pH value, the more acidic the soil, and conversely, the higher higher the pH value, the more alkaline the soil. The pH scale is logarithmic, so each pH unit change represents a tenfold change in acidity or alkalinity. For example, a soil with a pH of 6 is 10 times as acidic as one with a pH of 7 and a soil with a pH of 8 is 10 times as alkaline as one with a pH of 7 and 100 times as alkaline as one with a pH of 6.
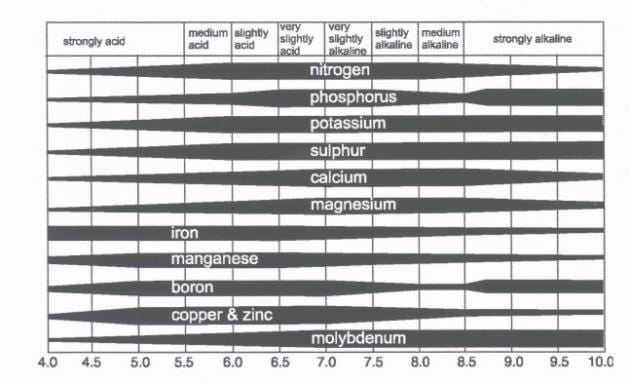
Note that the term alkaline refers to soil pH above 7.0; the term alkali is an older term for alkali is an older term for soils with excessive sodium. The soil reaction is important because it greatly influences nutrient availability, solubility of toxic ions and microbial activity. (see chart)
The maximum availability of the primary nutrients (N-P-K) is greatest at pH value between 6.5 and 7.0. The solubility of some phytotoxic elements, such as aluminum, increases at low pH values (below 5.5), which can reduce crop yields. Manganese may also become toxic at low pH levels. Sodium is generally excessive when the soil pH is above 8.5. Near neutral pH values favor the activity of many microorganisms responsible for essential biological activity in the soil.
Some crops (such as blueberry and strawberries) tend to grow better in slightly acidic soils. Consult our agronomy team for specific crop pH recommendations.
If you have high pH soils, elemental sulfur should be incorporated into the soil to help reduce the pH.
If you have lower pH soils, lime or dolomitic lime (if also low in magnesium) should be added to help raise the pH.
BENEFITS OF GYPSUM
Regular use of gypsum is essential to sustainability of most irrigated soils. Gypsum is a key ingredient for the maintenance of agriculture on many types of soils.
